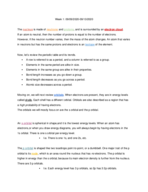PreparED Study Materials
CHEM 40551: General Chemistry I
School: University of St. Thomas
Number of Notes and Study Guides Available: 22
Notes
Study Guides
Chem 111 test #2 study guide (Engineering)
CHEM 40551
University of St. Thomas
9 pages | Summer 2015
Chem 111 test #2 study guide (Engineering)
CHEM 40551
University of St. Thomas
9 pages | Summer 2015
Chem 111 test #2 study guide (Engineering)
CHEM 40551
University of St. Thomas
9 pages | Summer 2015
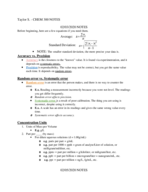
Chem 111 study guide exam #1 (Engineering)
CHEM 40551
University of St. Thomas
8 pages | Summer 2015
Videos
Finding NaOH Molarity: Titration of 0.200L SO?-Derived H?SO? Solution
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Determine the molarity of a NaOH solution through titration with sulfurous acid. Starting with the ideal gas equation we derive the concentration of a 0.200L SO?-derived H?SO? solution. Concluding with a molarity of 1.64 M for NaOH using calculated values.
Understanding Average Speed in Kinetic Molecular Theory
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
The kinetic molecular theory of gases describes the random motion and speed distribution of gas molecules in a container, and when considering various statements about the average speeds of these molecules, it's important to understand how they behave under different conditions.
Molecular Lewis Structures for CH?, CO?, H?Se, and NH?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video, we demonstrate the creation of Lewis structures for methane, carbon dioxide, hydrogen selenide, and ammonia, highlighting the arrangement of atoms and bonds in each molecule. The video emphasizes the importance of Lewis structures in visualizing atomic bonding and the presence of lone electron pairs.
Determining Formula Mass: Step-by-Step Approach Using Atomic Masses
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the method to determine the formula mass for various compounds. Learn about the importance of atomic masses and their role in compounds like Aluminium sulfate, Diphosphorus Trioxide, and more. Understand the step-by-step approach to accurately calculate molecular weights.
Converting Moles to Grams: Aluminum & Chlorine Atomic Mass Breakdown
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
"Explore the intricate world of atomic masses using aluminum and chlorine as examples. Understand the concept of 'mole' and its relevance in chemistry. Learn to calculate masses of moles for elements based on atomic weights.
Breaking Down Mixtures: Finding Mass Percent of Sodium Compounds
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Uncover the method to determine mass percentages of Sodium Bromide and Sodium Sulfate in a mixture using the given Sodium content. Walk through a step-by-step calculation leveraging molar masses to derive the composition. Discover the resulting mass percentages: 23.85% for Sodium Bromide and 76.15% for Sodium Sulfate