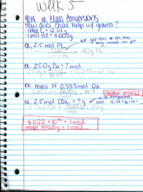PreparED Study Materials
CHE 242: Basic Biochemistry
School: Illinois State University
Number of Notes and Study Guides Available: 2
Notes
Videos
Physical vs. Chemical: Chlorine Reactions & Magnetism
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the differences between physical and chemical properties in substances. Learn some fundamentals of chemistry and understand how substances interact and change.
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Electron Configuration of Phosphorus: Decoding Atomic & Orbital Struct
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the intricacies of the electron configuration for phosphorus, from its atomic number to its orbital notation. Learn about unpaired electrons, energy levels, and the significance of inner-shell electrons. Understand how orbital structures define an element's properties.
Strong Base Comparison: H?S?, CH?O?, CH?OH, Cl?, CH?COO?, CH?CHClCOO
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the reactivity of different bases in this chemistry comparison. We'll analyze the strength of H?S?, CH?O?, CH?OH, Cl?, CH?COO?, and CH?CHClCOO? in various reactions.
Understanding Key Assumptions of Kinetic Molecular Theory in Detail!
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Dive into the Kinetic Molecular Theory of Matter understanding how atoms and molecules possess kinetic energy which we measure as temperature. Explore the five core assumptions including the constant random motion of gas particles and the relation between kinetic energy and temperature. Demystify the concepts of particle collisions intermolecular interactions and the universal behavior of gases at given temperatures
Lattice Energy: Always Positive; Hydration Energy: Always Negative
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video we clarify why lattice energy in solids is always a positive value due to the energy needed to separate ions making it an endothermic process. On the flip side hydration energy is always negative as it releases heat when ions dissolve in water making it an exothermic reaction. We use sodium chloride as a real-world example to illustrate these crucial thermodynamic concepts
Textbook Solutions (0)
Top Selling Study Tools
Che140 lecture notes 10.5 - 10.9 (Chemistry)
CHE 140
Illinois State University
5 pages | Summer 2015