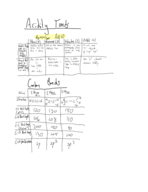
PreparED Study Materials
CHEM 130: Organic and Bio-Organic Chemistry Laboratory
School: Stanford University
Number of Notes and Study Guides Available: 1
Notes
Videos
Reversible Enzyme Inhibition: Good or Bad? Exploring Implications
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Delve into the complex world of enzymes and explore the implications of their reversible inhibition, shedding light on whether it's advantageous or detrimental to biological processes."
Determining Neutrons from Atomic & Mass Numbers: A Comprehensive Guide
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Understand the relationship between atomic number, mass number, and neutrons within atoms. This video breaks down the simple calculation to determine the number of neutrons for specific elements like Nickel, Uranium, Scandium, and Argon.
Molecule Count in 0.334 g of C?H?: Ethane Analysis
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This instructional video guides you through the process of determining the number of molecules in a given sample. It illustrates the conversion from grams to moles using the molar mass of the substance and then to molecules using Avogadro's number, providing a practical example with ethane (C?H?) to calculate approximately 6.67 x 10²¹ molecules in a 0.334 g sample.
Breaking Down Mixtures: Finding Mass Percent of Sodium Compounds
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Uncover the method to determine mass percentages of Sodium Bromide and Sodium Sulfate in a mixture using the given Sodium content. Walk through a step-by-step calculation leveraging molar masses to derive the composition. Discover the resulting mass percentages: 23.85% for Sodium Bromide and 76.15% for Sodium Sulfate
Nickel(II) Nitrate Solution Composition Analysis
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this analysis, we determine the composition of a solution prepared by dissolving 12.15g of nickel(II) nitrate in 175mL of water (density 1.00 g/mL). The mass percent of nickel(II) nitrate is found to be approximately 6.50%, and the mole fraction of nickel(II) ions in the solution is calculated as 0.00672.
Finding NaOH Molarity: Titration of 0.200L SO?-Derived H?SO? Solution
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Determine the molarity of a NaOH solution through titration with sulfurous acid. Starting with the ideal gas equation we derive the concentration of a 0.200L SO?-derived H?SO? solution. Concluding with a molarity of 1.64 M for NaOH using calculated values.