PreparED Study Materials
Videos
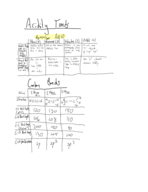
Finding NaOH Molarity: Titration of 0.200L SO?-Derived H?SO? Solution
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Determine the molarity of a NaOH solution through titration with sulfurous acid. Starting with the ideal gas equation we derive the concentration of a 0.200L SO?-derived H?SO? solution. Concluding with a molarity of 1.64 M for NaOH using calculated values.
Nickel(II) Nitrate Solution Composition Analysis
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this analysis, we determine the composition of a solution prepared by dissolving 12.15g of nickel(II) nitrate in 175mL of water (density 1.00 g/mL). The mass percent of nickel(II) nitrate is found to be approximately 6.50%, and the mole fraction of nickel(II) ions in the solution is calculated as 0.00672.
Reaction Rate Impact: Ethanol, Tert-Butyl Bromide & Temp
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the intricacies of reaction rates with varying concentrations of ethanol and tert-butyl bromide. Understand the principles of SN1 reactions and the impacts of temperature on rate acceleration. Grasp the core elements that influence and optimize chemical reactions in diverse applications.
Producing Ammonium Sulfate: Calculating the Required Ammonia
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the process to determine the quantity of ammonia (NH?) required to produce a specific amount of ammonium sulfate ((NH?)?SO?). Through a step-by-step explanation, learn the application of the balanced chemical equation and molar mass conversions. Transform theoretical chemistry into practical knowledge with this insightful guide.
Disulfur Decafluoride Disproportionation & Sulfur Oxidation States
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video explains the disproportionation reaction of disulfur decafluoride at 150°C. It teaches you to write a balanced equation for this reaction and give the oxidation state of S in each compound.
Calculate a Building's Age Using Nickel-63 Decay
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
The scenario involves an archaeologist analyzing iron in an old building containing nickel-63, which has a known half-life of 92 years. With only 0.78% of the original nickel-63 remaining, the goal is to estimate the year when this discovery was made. We delve into the calculations and principles of first-order decay reactions.