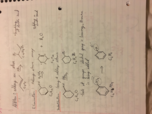PreparED Study Materials
CHEM 227: Organic Chemistry I
School: Texas A&M University
Number of Notes and Study Guides Available: 79
Notes
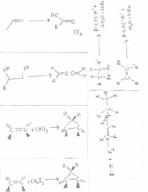
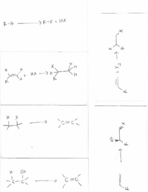
Alkene/alkyne: reactions and synthesis (Chemistry)
CHEM 227
Texas A&M University
8 pages | Fall 2015

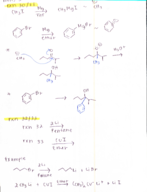
Organic chemistry 127 chapter 17 notes (Chemistry)
CHEM 227
Texas A&M University
6 pages | Fall 2015

Organic chemistry 127 chapter 10 notes (Chemistry)
CHEM 227
Texas A&M University
4 pages | Fall 2015

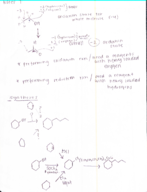
Organic chemistry 127 chapter 18 notes (Chemistry)
CHEM 227
Texas A&M University
6 pages | Fall 2015
Study Guides

Organic chemistry: exam 1 study review (Chemistry)
CHEM 227
Texas A&M University
6 pages | Fall 2015

Organic chemistry: exam 3 study guide (Chemistry)
CHEM 227
Texas A&M University
10 pages | Fall 2015
Videos
Crafting Ionic and Net Ionic Equations: Step-by-Step Guide with Exampl
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover how to craft balanced, complete ionic, and net ionic equations for various chemical reactions. Learn the significance of aqueous ions and solid state indicators in these equations.
Calculating MgO Mass from Oxygen Gas at STP
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video, the problem involves calculating the mass of magnesium oxide (MgO) produced when 14.8 liters of oxygen gas react with magnesium metal according to the chemical equation 2Mg + O2 -> 2MgO. The stoichiometric relationship is used to determine that 0.6607 moles of oxygen gas results in 1.3214 moles of MgO, with a final calculation yielding a mass of 53.25 grams of MgO formed during the reaction at Standard Temperature and Pressure (STP).
Understanding Chlorine's Oxidation States in Complex Ions: A Step-by-S
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore how to determine the oxidation states of Chlorine in various ions using fundamental rules in chemistry. Learn the importance of oxidation numbers in understanding chemical reactions and the art of balancing chemical equations.
Denver Pressure Conversion: Atmospheres mmHg psi & Pascals Explained
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Converting Denver's average pressure of 24.9 in. Hg into various units. This video provides clear steps to determine equivalents in millimeters of mercury atmospheres pounds per square inch and pascals. Understand the relationships and conversions between these common pressure units.
Volume of 49.8g HCl Gas at STP: Finding the Answer in Liters
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video guides viewers through the process of calculating the volume occupied by Hydrochloric acid gas or HCl at standard temperature and pressure. Using the molar mass of HCl and its properties as an ideal gas the tutorial comprehensively explains each step to determine the volume from a given mass at STP. A must-watch for understanding gas volume conversions in chemistry
Electronegativity & Acid Strength: Decoding Acid Formulas
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video discusses how the electronegativity of attached groups affects the strength of acids. Comparing pairs such as Chloroacetic vs Fluoroacetic and Acetic vs Nitroacetic acids, we highlight which is the stronger acid in each pair and why.