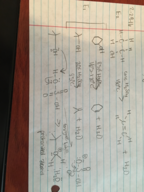PreparED Study Materials
CH 1244: Integrated Chemistry 1
School: Mississippi State University
Number of Notes and Study Guides Available: 0
Videos
Molecular Formulas: Lewis Structures for N2, HCN, CO2, C3H6, and More
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover Lewis structures for various molecular formulas, including N2, HCN, CO2, C3H6, and more. Explore their chemical bonding and structural arrangements.
Disulfur Decafluoride Disproportionation & Sulfur Oxidation States
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video explains the disproportionation reaction of disulfur decafluoride at 150°C. It teaches you to write a balanced equation for this reaction and give the oxidation state of S in each compound.
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Molecular Geometries Explained: From N?O to PF? - Shape and Structure
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the distinct geometries of molecules like N?O, SO?, H?S, and PF?. Unravel the difference between molecular and electron shapes. Master the basics of linear, bent, tetrahedral, and trigonal pyramidal structures.
What is the expected ground-state electron configuration for Te²??
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Join us in this illuminating video as we unravel the expected ground-state electron configuration for Te²?. We dive into the world of chemistry to explain the electron structure of this fascinating ion. Whether you're a student, a chemistry enthusiast, or simply curious about the periodic table, this video provides a clear and comprehensive breakdown of the electron configuration for Te²?. Discover the secrets of electron arrangement and explore the intriguing properties of tellurium in its doub
Mole Count in Elemental Samples (Zn, Ar, Ta, Li)
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video outlines the process of calculating the number of moles in various elemental samples. It begins by explaining the relationship between molar mass and atomic weight. Each sample's mass is divided by the respective element's molar mass to find the number of moles. It provides detailed calculations for four elemental samples: zinc (Zn), argon (Ar), tantalum (Ta), and lithium (Li), converting results to scientific notation where necessary. The script demonstrates how to determine the mole