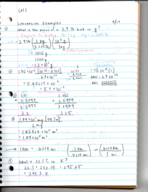PreparED Study Materials
CHEM 467: Biochemistry of Disease
School: Colorado State University
Number of Notes and Study Guides Available: 0
Videos
Calculating Silver Density from Weight and Water Displacement
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how to find the density of silver using weight and water displacement. A step-by-step guide to a common laboratory technique.
Homogeneous vs Heterogeneous Mixtures: Examples & Classification Expla
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the distinction between homogeneous and heterogeneous mixtures. Through real-world examples like baby oil, chocolate chip cookies, and wine, understand their uniformity, phases, and how components are distributed.
Calculating Ksp for Ca3(PO4)2 at 25°C: Step-by-Step Guide
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Unravel the concepts of 'Molar Solubility' and 'Solubility Product' to gauge salt solubility. Explore how Calcium Phosphate dissociates in solutions and calculate its unique Ksp value.
Determining Mass Percent Concentration of Solutions
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This instructional video demonstrates the process of calculating the mass percent of various solutions, using the formula "mass of solute divided by mass of solution, all multiplied by 100%." It provides step-by-step calculations for three different solutions, revealing the concentration of each solute in terms of mass percent, offering a practical understanding of quantifying solute proportions in solutions.
Electron Configuration of Phosphorus: Decoding Atomic & Orbital Struct
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the intricacies of the electron configuration for phosphorus, from its atomic number to its orbital notation. Learn about unpaired electrons, energy levels, and the significance of inner-shell electrons. Understand how orbital structures define an element's properties.
Identify the 3' and 5' ends of the DNA segment AGTCAT
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video discusses the fundamental concept of DNA structure, focusing on the identification of the 3' and 5' ends of a DNA segment, specifically highlighting the sequence AGTCAT and its importance in understanding DNA strand direction for processes like replication and transcription.
Textbook Solutions (0)
Top Selling Study Tools

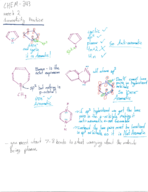
Chem 343 week 1 notes (8/22-8/26) (Chemistry)
CHEM 343
Colorado State University
7 pages | Fall 2015

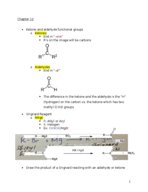
Organic chemistry week 4 notes (Chemistry)
CHEM 341
Colorado State University
11 pages | Spring 2016