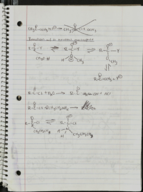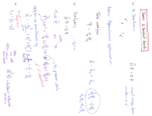PreparED Study Materials
CHEM 2760: Organic Chemistry 2760
School: East Carolina University
Number of Notes and Study Guides Available: 86
Notes
Study Guides
Videos
Differential DNA Damage Responses in Prokaryotes and Eukaryotes
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore unique strategies employed by prokaryotes and discover if they possess exclusive mechanisms for handling extreme DNA damage that elude eukaryotes."
Mastering Ionic and Net Ionic Equations for Varied Chemical Reactions
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the art of writing balanced, complete ionic, and net ionic equations for various chemical reactions. Learn the nuances that differentiate between these equations and watch them applied to diverse chemical reactants.
Animal and Plant Energy Storage: Fats and Oils Explained
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video delves into the fascinating world of energy storage in animals and plants, offering insights into why fats are the primary energy storage compound in animals (except muscles) and why plants opt for alternative strategies instead of using fats and oils.
Calculating Vapor Pressure & Its Lowering: Urea in Water at 35°C
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the intriguing process of determining the vapor pressure of a urea-water solution at 35°C. Using Raoult's law we'll calculate the mole fraction of water and urea leading to the solution's vapor pressure. Learn how the solution's composition affects vapor-pressure lowering
Mole Count in Elemental Samples (Zn, Ar, Ta, Li)
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video outlines the process of calculating the number of moles in various elemental samples. It begins by explaining the relationship between molar mass and atomic weight. Each sample's mass is divided by the respective element's molar mass to find the number of moles. It provides detailed calculations for four elemental samples: zinc (Zn), argon (Ar), tantalum (Ta), and lithium (Li), converting results to scientific notation where necessary. The script demonstrates how to determine the mole
Producing Ammonium Sulfate: Calculating the Required Ammonia
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the process to determine the quantity of ammonia (NH?) required to produce a specific amount of ammonium sulfate ((NH?)?SO?). Through a step-by-step explanation, learn the application of the balanced chemical equation and molar mass conversions. Transform theoretical chemistry into practical knowledge with this insightful guide.