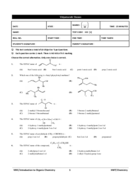PreparED Study Materials
CHEM 1031: The Science of Change
School: Auburn University
Number of Notes and Study Guides Available: 13
Notes
Videos
Chemical Combo: How N2 and F2 Produce Nitrogen Trifluoride Explained!
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the chemical combination of nitrogen and fluorine to produce nitrogen trifluoride. Learn how to determine the limiting reactant and calculate the theoretical yield using stoichiometry principles.
Orthorhombic Unit Cell: Decoding Nickel Sulfate's Crystal Density
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
The video offers an insightful look into the orthorhombic unit cell, a unique crystal lattice structure. By exploring nickel sulfate's unit cell and its specific dimensions, we determine both the number of formula units and the density of this crystalline structure.
Dimethylamine vs. Trimethylamine: Structures & Boiling Points
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Uncover the science behind boiling points and molecular interactions in this video. Explore why Dimethylamine boils at a higher temperature than Trimethylamine, despite having a lower molecular weight, and gain insights into the role of molecular structures in determining physical properties.
Neon Gas Pressure Change in 0.75-L Cylinder
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this problem, we are tasked with determining the pressure of neon gas in a 0.75-L cylinder at 30°C after compressing it from a 2.50-L sample at 0.00°C and 1.00 atm using the combined gas law equation, with step-by-step calculations leading to a final pressure value of approximately 3.699 atm.
Electron Configuration of Phosphorus: Decoding Atomic & Orbital Struct
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the intricacies of the electron configuration for phosphorus, from its atomic number to its orbital notation. Learn about unpaired electrons, energy levels, and the significance of inner-shell electrons. Understand how orbital structures define an element's properties.
Volume Calculation for Acetone, Olive Oil, and Chloroform
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video, the presenter demonstrates the process of calculating the volume of 125g for three different liquids—Acetone, Olive oil, and Chloroform—using the formula "Volume = Mass / Density," providing step-by-step solutions for each liquid, ultimately revealing their respective volumes. This instructional video guides viewers through the calculations and showcases the outcome for each liquid's volume.