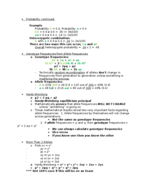PreparED Study Materials
CHM 2210: Organic Chemistry 1
School: Florida Atlantic University
Number of Notes and Study Guides Available: 20
Notes
Study Guides
Videos
Understanding Chlorine's Oxidation States in Complex Ions: A Step-by-S
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore how to determine the oxidation states of Chlorine in various ions using fundamental rules in chemistry. Learn the importance of oxidation numbers in understanding chemical reactions and the art of balancing chemical equations.
Chromium Oxides: Acidity Comparison in Water
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Compare the acidity of chromium trioxide (CrO?) and chromium (II) oxide (CrO) in water. Chromium trioxide forms strong chromic acid increasing hydronium ions, while chromium (II) oxide has weaker oxidizing properties, resulting in a less acidic solution.
Determining Base Concentration Using KHP: Indicator Choice Solution
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover how to choose the right indicator for a titration involving Potassium hydrogen phthalate (KHP) and a strong base. Grasp the calculations from molar mass to pH and understand why Thymolphthalein is the ideal choice for a change near pH 8.95.
Metric Conversions: cm to m, g to kg, cm to mm, mL to L
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Master metric unit conversions with step-by-step examples, including cm to meters, g to kilograms, cm to millimeters, and mL to liters.
Reversible Enzyme Inhibition: Good or Bad? Exploring Implications
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Delve into the complex world of enzymes and explore the implications of their reversible inhibition, shedding light on whether it's advantageous or detrimental to biological processes."
Number of molecules in 3.5g H?O, 56.1g N?, 89g CCl?, 19g C?H??O?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video shows how to determine the number of molecules in a given sample by utilizing the concept of moles, Avogadro's number (6.022 x 10²³), and the molar mass of the substance. It provides step-by-step calculations for different substances, converting mass to the number of molecules and highlighting the versatile application of this approach.