PreparED Study Materials
CHEM 1213: Chem 1
School: Mississippi State University
Number of Notes and Study Guides Available: 2
Study Guides
Videos
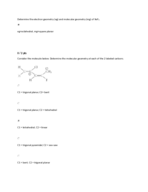
Lewis Structures with Atom Connections & Charges
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how to create Lewis structures for various molecules and assign charges where needed. Understand the molecular connectivity and electron distribution.
Tailors' Seam Measurements: Precision and Accuracy Analysis
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
An in-depth analysis of the precision and accuracy in seam measurements by three apprentice tailors: X, Y, and Z, as they measure a pair of trousers. Understand how their measurements compare to the true length of 32.0 inches.
Understanding Key Assumptions of Kinetic Molecular Theory in Detail!
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Dive into the Kinetic Molecular Theory of Matter understanding how atoms and molecules possess kinetic energy which we measure as temperature. Explore the five core assumptions including the constant random motion of gas particles and the relation between kinetic energy and temperature. Demystify the concepts of particle collisions intermolecular interactions and the universal behavior of gases at given temperatures
Understanding Polar & Nonpolar Molecules: From Diazene to Nitrogen Dio
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the properties of various molecules like diazene, hydrogen peroxide, and carbon tetrafluoride. Understand the influence of molecular shape and electronegativity on polarity. Discover why some are polar while others are nonpolar based on their structure.
Drawing Open-Chain Structures: A Visual Guide to Four Sugars
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how to draw open-chain structures for various sugars, from ketotetrose to amino sugars. This video offers visual demonstrations of ketopentose, deoxyaldohexose, and more, highlighting key molecular differences.
Vaporization vs. Condensation: Heat Absorption & Release
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the contrasting processes of vaporization and condensation and their thermal nature. Understand why boiling is endothermic, absorbing heat, while condensation is exothermic, releasing heat, and how these processes impact ?H values.