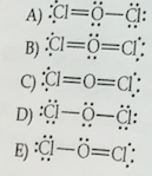PreparED Study Materials
CH 405: Medicinal Chemistry
School: University of Alabama - Tuscaloosa
Number of Notes and Study Guides Available: 21
Notes
Pharmacokinetics, pt. 1 (Chemistry)
CH 405
University of Alabama - Tuscaloosa
15 pages | Spring 2015
Study Guides
Videos
Deriving the Perfect Gas Equation from Gas Laws
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how the ideal gas equation is derived from Boyle's law, Charles’ law, and Avogadro's principle, unraveling the fundamentals of gas behavior.
Molarity Calculations for Various Solutions: CH?OH, CaCl?, C??H?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how to calculate the molarity of different solutions containing methanol, calcium chloride, and naphthalene. Essential for chemistry enthusiasts.
Predicting Precipitates: Al(NO?)? & NaOH Reaction Explained
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Unlock the secrets of precipitation reactions as we mix Aluminium nitrate and Sodium hydroxide. Discover how Al(NO?)? and NaOH interact forming the insoluble Aluminium hydroxide Al(OH)?. Break down the net ionic equation to highlight only the active participants.
Physical vs Chemical Changes: Understanding Reactions & Transformation
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the differences between physical and chemical changes using common examples. Understand how sugar reacts in various scenarios, and how metals undergo changes, either maintaining their substance or forming new compounds.
Differential DNA Damage Responses in Prokaryotes and Eukaryotes
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore unique strategies employed by prokaryotes and discover if they possess exclusive mechanisms for handling extreme DNA damage that elude eukaryotes."
Sketching Lewis Structures: Radical Species ClF? and BrO? Explained
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video elucidates the method to sketch Lewis structures for the radical species ClF? and BrO?. Highlighting the distinguishing feature of an unpaired electron, we illustrate the bond arrangements and lone pairs that define these unique chemical entities.