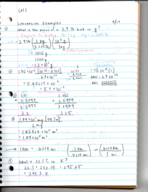PreparED Study Materials
CHEM 62138: General Chemistry I
School: Colorado State University
Number of Notes and Study Guides Available: 2
Notes
Videos
Chemical Formulas: CaI?, N?O?, SiO?, ZnCl? for Atom Ratios
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Unlock the world of chemical formulas and their atom ratios. Learn to write compounds like CaI?, N?O?, SiO?, and ZnCl? in this chemistry tutorial.
Where in the cell does fatty acid catabolism take place?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video explores the process of fatty acid catabolism, highlighting its occurrence in the mitochondria, the crucial role of mitochondria in energy generation, and the metabolic reactions involved without revealing specific details or conclusions.
What makes bubbles when water boils?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
When water boils, bubbles rise to the surface, but what's in these bubbles? Let's evaluate the options. Option d, water vapor, is correct; the bubbles during boiling are primarily composed of water vapor, the gaseous state of water.
Understanding STP: Calculating Gas Volumes at 0°C and 1 ATM
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the fundamental concept of Standard Temperature and Pressure (STP) and its relevance in determining the volume of different gases. Learn to calculate gas volumes at STP using real-life examples of Chlorine, Nitrogen, Helium, and Methane. Grasp essential chemistry concepts seamlessly with practical demonstrations.
Why is Chloromethane Polar but Methane Nonpolar? Decoding Molecule Sha
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the polar nature of Chloromethane and the nonpolar characteristics of Methane. Understand the impact of electronegativity differences and bond types. Deciphering molecular polarity through the lens of tetrahedral structures.
Understanding Reaction Rate Decrease Over Time
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This explanation delves into the phenomenon where the reaction rate decreases over time due to the decrease in reactant concentration, resulting in fewer successful collisions and reactions, without revealing specific examples or findings.
Textbook Solutions (0)
Top Selling Study Tools

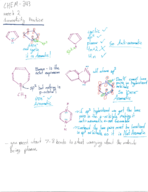
Chem 343 week 1 notes (8/22-8/26) (Chemistry)
CHEM 343
Colorado State University
7 pages | Fall 2015

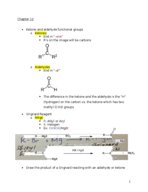
Organic chemistry week 4 notes (Chemistry)
CHEM 341
Colorado State University
11 pages | Spring 2016