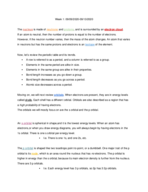PreparED Study Materials
CHEM 201: Organic Chemistry 1
School: University of St. Thomas
Number of Notes and Study Guides Available: 4
Notes
Videos
Calculating MgO Mass from Oxygen Gas at STP
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video, the problem involves calculating the mass of magnesium oxide (MgO) produced when 14.8 liters of oxygen gas react with magnesium metal according to the chemical equation 2Mg + O2 -> 2MgO. The stoichiometric relationship is used to determine that 0.6607 moles of oxygen gas results in 1.3214 moles of MgO, with a final calculation yielding a mass of 53.25 grams of MgO formed during the reaction at Standard Temperature and Pressure (STP).
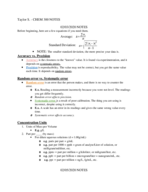
Calculating Percent Composition: Using Molecular Formulas & Atomic Mas
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Understand the process of calculating percent composition using the molecular formula. Grasp the methodology with compounds like C2H4O2, CH2O2, and more. Discover how atomic masses influence the mass percent of elements in various compounds.
What are the ground-state electron configurations of Ge, Fe, Zn, Ni, W
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video provides ground-state electron configurations for various elements, including germanium, iron, zinc, nickel, tungsten, and thallium, based on their atomic numbers.
Dimethylamine vs. Trimethylamine: Structures & Boiling Points
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Uncover the science behind boiling points and molecular interactions in this video. Explore why Dimethylamine boils at a higher temperature than Trimethylamine, despite having a lower molecular weight, and gain insights into the role of molecular structures in determining physical properties.
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Why is it necessary that protein molecules be enormous?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This discussion explores the importance of the size of protein molecules, highlighting their three-dimensional structures, diverse functions, and the role of large size in enabling structural motifs, functional regions, and stability without revealing specific examples or findings.