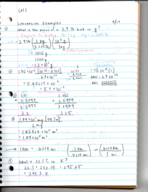PreparED Study Materials
CHEM 103: Chemistry in Context (GT-SC2)
School: Colorado State University
Number of Notes and Study Guides Available: 4
Notes
Videos
Strontium: Deep Dive into Its Isotopes & Atomic Structure
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the step-by-step process of Barium Sulfate formation from the reaction of Barium Chloride with Sodium Sulfate. Learn about molar mass calculations stoichiometry and yield predictions.
Neon Gas Pressure Change in 0.75-L Cylinder
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this problem, we are tasked with determining the pressure of neon gas in a 0.75-L cylinder at 30°C after compressing it from a 2.50-L sample at 0.00°C and 1.00 atm using the combined gas law equation, with step-by-step calculations leading to a final pressure value of approximately 3.699 atm.
Speed of Light Explained: Earth, Billions of Km & the Concept of Light
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the incredible speed of light as it zips 7.5 times around Earth in just a second! Marvel at its daily and yearly travels, equating to billions and trillions of kilometers. Dive deeper into the concept of a 'light year' and its significance in astronomical measurements.
Comparing Structural Features of Sphingomyelins and Glycolipids
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video explores the structural and functional differences between sphingomyelins and glycolipids, two types of lipids found in cell membranes, highlighting their commonalities in backbone and fatty acid composition and their distinctive roles in cell structure and recognition.
Why is a short RNA primer needed for replication?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Unlocking the Mystery of Short RNA Primers in DNA Replication: Discover the essential role they play and why they are a crucial component in the replication process. Join us in this video as we delve into the fascinating world of molecular biology and DNA replication.
Endothermic Rxn: Ba(OH)2·8H2O & NH4NO3 Heat Dynamics
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the endothermic reaction between Ba(OH)2·8H2O and NH4NO3 observing its impact on surroundings like the freezing water. Discover why touching the beaker feels cold and learn about heat transfer in such chemical reactions. This video is an insightful demonstration of heat absorption in chemical reactions.
Textbook Solutions (0)
Top Selling Study Tools

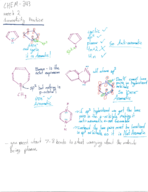
Chem 343 week 1 notes (8/22-8/26) (Chemistry)
CHEM 343
Colorado State University
7 pages | Fall 2015

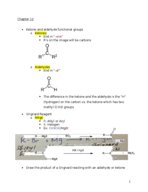
Organic chemistry week 4 notes (Chemistry)
CHEM 341
Colorado State University
11 pages | Spring 2016