PreparED Study Materials
Notes
Videos
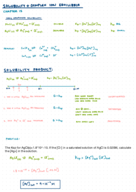
Ethylene Glycol: Mass and Volume Calculations
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore how to calculate the mass and volume of ethylene glycol, a common antifreeze, using its density. Understand the essentials of density-related calculations.
Homogeneous vs Heterogeneous Mixtures: Examples & Classification Expla
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the distinction between homogeneous and heterogeneous mixtures. Through real-world examples like baby oil, chocolate chip cookies, and wine, understand their uniformity, phases, and how components are distributed.
Volume of 49.8g HCl Gas at STP: Finding the Answer in Liters
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video guides viewers through the process of calculating the volume occupied by Hydrochloric acid gas or HCl at standard temperature and pressure. Using the molar mass of HCl and its properties as an ideal gas the tutorial comprehensively explains each step to determine the volume from a given mass at STP. A must-watch for understanding gas volume conversions in chemistry
Neon Gas Pressure Change in 0.75-L Cylinder
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this problem, we are tasked with determining the pressure of neon gas in a 0.75-L cylinder at 30°C after compressing it from a 2.50-L sample at 0.00°C and 1.00 atm using the combined gas law equation, with step-by-step calculations leading to a final pressure value of approximately 3.699 atm.
Unraveling Hydrocarbon Structures: Decoding Degrees of Unsaturation
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the concept of the degree of unsaturation in hydrocarbons. Understand its importance in revealing double bonds, triple bonds, or rings. Learn through examples like limonene, squalene, and lycopene.
What is a propeller twist?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Unravel the concept of propeller twist and its role in aviation and marine propulsion. We'll delve into the science and engineering behind this intriguing phenomenon, shedding light on its applications and significance.
Textbook Solutions (0)
Top Selling Study Tools

Chem 1332, chapter 10, 11 (week 2) (Chemistry)
CHEM 14833
University of Houston
12 pages | Fall 2016