PreparED Study Materials
Notes
Videos
Intermolecular Forces in HCl, F?, and C?H??
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the fascinating world of intermolecular forces in this brief video, featuring HCl, F?, and C?H?? molecules. Learn about the bonds that hold these substances together!
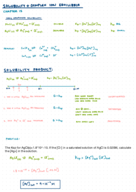
Chemical Formulas: CaI?, N?O?, SiO?, ZnCl? for Atom Ratios
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Unlock the world of chemical formulas and their atom ratios. Learn to write compounds like CaI?, N?O?, SiO?, and ZnCl? in this chemistry tutorial.
Key Biochemical Concepts: Polymer, Protein, Nucleic Acid, Catalysis, G
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In biochemistry, polymers are essential macromolecules, including carbohydrates, lipids, proteins, and nucleic acids, each adopting specific shapes and performing vital functions within cells, with proteins serving diverse roles, nucleic acids carrying genetic information, catalysis expediting reactions, and the genetic code enabling protein synthesis.
Determining Bond Types: Ionic, Polar Covalent, or Covalent in Chemical
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This discussion revolves around the classification of chemical bonds as ionic, polar covalent, or covalent based on the electronegativity difference between the atoms involved. The general criteria for this classification are explained: a difference greater than 1.7 indicates an ionic bond, between 0.5 and 1.7 signifies a polar covalent bond, and less than 0.5 designates a covalent bond. It then applies these criteria to four specific examples, including the CC bond in ethane (H?CCH?), the KI bo
Perfecting Perbromate Ion: Achieving Zero Formal Charges
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how to create a Lewis structure for the perbromate ion, ensuring all atoms achieve zero formal charges. Follow the step-by-step guide to understand the bonding and electron distribution in this chemical compound.
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Textbook Solutions (0)
Top Selling Study Tools

Chem 1332, chapter 10, 11 (week 2) (Chemistry)
CHEM 14833
University of Houston
12 pages | Fall 2016