PreparED Study Materials
CHEM 2510: Organic Chemistry
School: University of Houston
Number of Notes and Study Guides Available: 0
Videos
Crafting Ionic and Net Ionic Equations: Step-by-Step Guide with Exampl
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover how to craft balanced, complete ionic, and net ionic equations for various chemical reactions. Learn the significance of aqueous ions and solid state indicators in these equations.
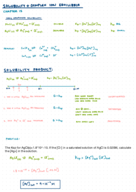
Determining Moles of Released Ions in Dissolution Reactions
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
When an ionic compound dissolves in water, it undergoes dissociation into its constituent ions. The total moles of ions released is determined by adding up the moles of each ion generated during this dissociation process. In the case of (a) disodium hydrogen phosphate (Na?HPO?), it dissociates into two sodium ions and one hydrogen phosphate ion. For (b) copper(II) sulfate pentahydrate (CuSO? · 5H?O), it dissociates into one copper ion and one sulfate ion. In (c), nickel(II) chloride (NiCl?) diss
Barium Chloride & Sodium Sulfate: Calculating BaSO? Yield
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Dive into the chemical reaction between Barium Chloride and Sodium Sulfate. Through step-by-step calculations, discover how to determine the resultant mass of Barium Sulfate. Conclude with a real-world example, highlighting the precise amount formed.
Glucose Polymer with Alternating Glycosidic Linkages: Structure and Fu
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the fascinating discovery of a glucose polymer with alternating glycosidic linkages. We'll visualize its Haworth projection and investigate its potential role in organisms, pondering whether it primarily serves a structural or energy storage function. Additionally, we'll discuss which types of organisms might utilize this unique polysaccharide as a potential food source.
Calculating MgO Mass from Oxygen Gas at STP
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video, the problem involves calculating the mass of magnesium oxide (MgO) produced when 14.8 liters of oxygen gas react with magnesium metal according to the chemical equation 2Mg + O2 -> 2MgO. The stoichiometric relationship is used to determine that 0.6607 moles of oxygen gas results in 1.3214 moles of MgO, with a final calculation yielding a mass of 53.25 grams of MgO formed during the reaction at Standard Temperature and Pressure (STP).
Ether Structures: Ethyl to Dipentyl Varieties Drawn
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the chemical structures of four distinct ethers: ethyl propyl ether dibutyl ether methyl hexyl ether and dipentyl ether. Understand the role of ether groups which involve an oxygen atom connected to two alkyl or aryl groups.
Textbook Solutions (0)
Top Selling Study Tools

Chem 1332, chapter 10, 11 (week 2) (Chemistry)
CHEM 14833
University of Houston
12 pages | Fall 2016