PreparED Study Materials
CHEM 5: History of Rock and Roll
School: University of Houston
Number of Notes and Study Guides Available: 2
Notes
Videos
Using Avogadro's Number: Calculating Moles of Aluminum & Iron Atoms
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how to determine the number of moles in a given set of atoms using Avogadro's number. This tutorial breaks down the calculation for Aluminum and Iron, simplifying the concept for better understanding.
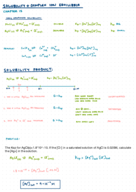
Does Cesium Bromide & Hydrogen Iodide in Water Conduct Electricity?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the conductivity of aqueous solutions: Cesium bromide and hydrogen iodide. Understand their ionic nature and how it leads to the presence of charged ions enabling electricity conduction
0.800g Match to SO?: Volume Calculation at 725 Torr & 32°C!
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Detailed walkthrough of calculating sulfur dioxide gas volume produced from burning tetraphosphorus trisulfide. Utilizes stoichiometry and the Ideal Gas Law to derive results under specific conditions. Step-by-step guide for understanding chemical reactions and gas volume calculations
Iron Bar: Mass Change and Rust Formation
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn about mass changes in an iron bar when exposed to moist air, including the formation of rust (Fe?O?) and how to calculate the final mass.
Hypothesis to Theory: The Evolution of Scientific Understanding
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Delve into the journey of scientific ideas as we discuss how a hypothesis can evolve into a theory, and whether a theory can ever become a law. Uncover the distinctions and connections between these fundamental elements of scientific understanding in this illuminating video.
Finding NaOH Molarity: Titration of 0.200L SO?-Derived H?SO? Solution
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Determine the molarity of a NaOH solution through titration with sulfurous acid. Starting with the ideal gas equation we derive the concentration of a 0.200L SO?-derived H?SO? solution. Concluding with a molarity of 1.64 M for NaOH using calculated values.
Textbook Solutions (0)
Top Selling Study Tools

Chem 1332, chapter 10, 11 (week 2) (Chemistry)
CHEM 14833
University of Houston
12 pages | Fall 2016