PreparED Study Materials
CEM 141: General Chemistry
School: Michigan State University
Number of Notes and Study Guides Available: 61
Notes
Study Guides
Videos
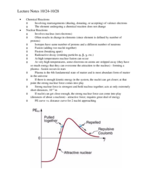
Calculating Percent Composition: Using Molecular Formulas & Atomic Mas
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Understand the process of calculating percent composition using the molecular formula. Grasp the methodology with compounds like C2H4O2, CH2O2, and more. Discover how atomic masses influence the mass percent of elements in various compounds.
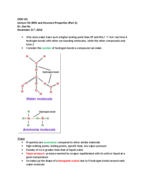
Comparing Masses: Analyzing Water, Ethanol, Chloroform & Balsa Wood
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the relationship between mass, density, and volume using real-life examples, including water, ethanol, chloroform, and balsa wood. Learn the process of calculating mass and uncover which substance has the highest mass based on given conditions.
Comparing Bond Types: Ethane's Nonpolarity vs. Fluoromethane's Ionic L
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Examine the nature of bonds in methylamine, ethane, fluoromethane, and methanol. Identify ethane's nonpolar covalent bond and understand why fluoromethane's bond leans toward the ionic spectrum. Grasp essential concepts in molecular bonding with this deep analysis.
Understanding Microwaves: Boiling Water's Time and Photon Count
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video breaks down the process of calculating the time and number of photons required to boil water in a microwave. Using given power, wavelength, and water specifics, we determine both energy needs and photon count.
Why geometric isomerism is not possible in alkynes
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video provides insights into the concept of geometric isomerism in organic chemistry and explains why alkynes, which contain carbon-carbon triple bonds and adopt a linear structure, cannot exhibit this form of isomerism, unlike alkenes with double bonds that restrict rotation.
Decoding Compound Names: What's HNO2(aq) Called in Chemistry
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the process of naming acids based on their anions and oxidation states. This video breaks down the correct naming of HNO2(aq) by distinguishing its anion and determining its place in the world of mineral acids.