PreparED Study Materials
CHEM 1213: Recitation for CHEM 1211
School: Northeastern University
Number of Notes and Study Guides Available: 1
Notes
Videos
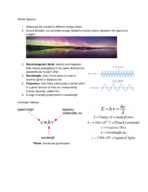
Strontium: Deep Dive into Its Isotopes & Atomic Structure
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the step-by-step process of Barium Sulfate formation from the reaction of Barium Chloride with Sodium Sulfate. Learn about molar mass calculations stoichiometry and yield predictions.
Calculating the Mass of CCl4: Using Density and Volume Insights
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn the method to calculate the mass of a substance using its density and volume. This video demonstrates the practical application of density in determining the mass of carbon tetrachloride (CCl4) from a given volume.
Converting Pressure Units to Atmospheres: mmHg, bars, Torr, kPa
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this instructional video, we explore the conversion of various pressure units into atmospheres, covering mmHg, bars, Torr, and kPa. The process involves utilizing conversion factors to ensure accurate results for each unit. By the end, you'll have equivalent pressures in atmospheres for the given values, enhancing your understanding of pressure conversions.
Determining Base Concentration Using KHP: Indicator Choice Solution
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover how to choose the right indicator for a titration involving Potassium hydrogen phthalate (KHP) and a strong base. Grasp the calculations from molar mass to pH and understand why Thymolphthalein is the ideal choice for a change near pH 8.95.
Unveiling the Water-to-Salt Ratio in Barium Chloride Hydrate
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the process of determining the number of water molecules in a barium chloride hydrate. Through a reaction with sulfuric acid, the video highlights how to derive the formation of barium sulfate and its mass calculations. A comprehensive chemical analysis leads to finding the elusive 'x' value, revealing the water-to-salt ratio.
Classifying Substances: From Pure Elements to Mixtures Explained
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Classify substances into various categories: element compound homogeneous mixture and heterogeneous mixture. This video breaks down the fundamental distinctions between pure substances and mixtures illustrating with real-world examples like urine pure water a Snickers™ bar and soil. A concise guide for understanding the essence of material classification in chemistry.