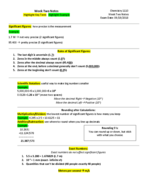
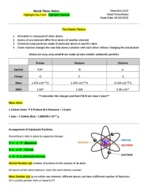
PreparED Study Materials
CHEM 1110: Elementary Chemistry
School: Southern Utah University
Number of Notes and Study Guides Available: 6
Notes
Study Guides
Videos
SN2 Rate: Doubling NaOH & 1-Bromopropane Effects
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
"Explore how altering concentrations and volume impacts the rate of an SN2 reaction between 1-bromopropane and sodium hydroxide. Learn the rate equation that describes this reaction and see real examples of how rate changes with concentration and volume adjustments
Why is it necessary that protein molecules be enormous?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This discussion explores the importance of the size of protein molecules, highlighting their three-dimensional structures, diverse functions, and the role of large size in enabling structural motifs, functional regions, and stability without revealing specific examples or findings.
Understanding Reaction Rate Decrease Over Time
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This explanation delves into the phenomenon where the reaction rate decreases over time due to the decrease in reactant concentration, resulting in fewer successful collisions and reactions, without revealing specific examples or findings.
Decoding Ions: Al³?, S²?, I?, Ag? & Their Electron-Proton Dynamics
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Break down the mysteries of ions like Al³?, S²?, I?, and Ag?. Discover how the electron count impacts ion charge by exploring Aluminium, Sulfur, Iodine, and Silver. Enhance your understanding of protons, electrons, and their relationship in ions.
Heat Absorption: Lead Weight's Final Temperature
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Explore the process of heat absorption and discover how to calculate the final temperature of a lead fishing weight after absorbing a specific amount of heat.
Weight% of Ba, K, Cl in Heated BaCl2·2H2O & KCl Mix
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video we explore the process of calculating the weight percent of barium potassium and chlorine in a mixed sample of barium chloride dihydrate and potassium chloride. Through a step-by-step approach we examine the changes in sample weight after heating to 160°C breaking down the calculations for each elemental content.