PreparED Study Materials
CHEM 1332: Fundamentals of Chemistry 2
School: University of Houston
Number of Notes and Study Guides Available: 77
Notes

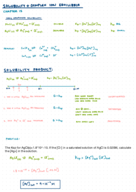
Chapter 17 notes: electrochemistry (Chemistry)
CHEM 1332
University of Houston
5 pages | Spring 2015
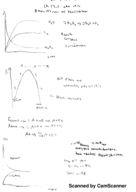
Chapter 20 lecture and video notes (Chemistry)
CHEM 1332
University of Houston
3 pages | Spring 2015
Study Guides
Videos
Calculating Silver Density from Weight and Water Displacement
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Learn how to find the density of silver using weight and water displacement. A step-by-step guide to a common laboratory technique.
pAg in KI & AgNO3 Titration: Exploring 39mL & 44.30mL Volumes
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Exploring the titration of 25.00 mL of 0.08230 M KI with 0.05110 M AgNO3. This video breaks down the calculation process for the potential of silver ion at various volumes of added AgNO3: 39.00 mL equivalence point and 44.30 mL. With clear steps and using the solubility product constant understand how the potential of silver ion is determined for each scenario.
Mass of Solutions: 12g Sucrose in 4.1%, 3.2%, & 12.5% Cases
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Determining the total mass of sucrose solutions at varying concentrations: 4.1% 3.2% and 12.5%. Learn step-by-step calculations to find solution masses containing 12 grams of sucrose. Results: ~292.68g ~375g and 96g for each concentration respectively.
The steps in the electron transport chain from NADH to oxygen
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
The electron transport chain (ETC) is a critical process in cellular respiration and photosynthesis, involving the transfer of electrons and protons through a series of protein complexes and molecules to create an electrochemical gradient, ultimately producing ATP with molecular oxygen as the final electron acceptor.
Electrons in outer shell of: Potassium, Calcium, Aluminum
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
This video explains the importance of the number of valence electrons in an element's chemical behavior and demonstrates how to determine the valence electrons for potassium, calcium, and aluminum based on their positions in the periodic table. Valence electrons dictate an element's reactivity with other elements, making this knowledge essential for understanding chemical reactions.
Strontium: Deep Dive into Its Isotopes & Atomic Structure
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Discover the step-by-step process of Barium Sulfate formation from the reaction of Barium Chloride with Sodium Sulfate. Learn about molar mass calculations stoichiometry and yield predictions.
Textbook Solutions (0)
Top Selling Study Tools

Chem 1332, chapter 10, 11 (week 2) (Chemistry)
CHEM 14833
University of Houston
12 pages | Fall 2016