PreparED Study Materials
CHEM 1111: Fundamentals of Chm Lab
School: University of Houston
Number of Notes and Study Guides Available: 0
Videos
Mole Fractions, Partial Pressures, and Total Pressure Calculation
Want To Learn More? To watch the entire video and ALL of the videos in the series:
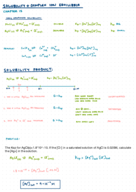
full solution
Explore the calculations for mole fractions, partial pressures, and total pressure in a 22.4 dm³ vessel with H? and N? at 273.15 K.
Quantifying Atoms Molecules & Moles: A Comprehensive Chemistry Guide
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Grasp the fundamental concept of moles in chemistry by equating it to the universally understood notion of a dozen. This video breaks down the usage of Avogadro's constant demonstrating calculations from moles to particles and vice versa. Using real-world examples like Carbon atoms Sulfur Dioxide molecules and Iron atoms viewers gain a clear understanding of moles and particle conversions
BrF?, ClF?, & IF? Lewis Structures: Which Don't Obey Octet Rule?
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Unravel the Lewis structures of BrF?, ClF?, and IF? and their relationship with the octet rule. Examine how fluorine bonds with bromine, chlorine, and iodine. Identify molecules that deviate from the standard octet distribution.
Evolutionary Implications of Cytochromes vs. Hemoglobin and Myoglobin
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
In this video, we delve into the fascinating world of evolutionary adaptations in heme-containing proteins.
Mass Calc: C?F, MgF?, CS? & SO? Explained
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Master the conversion of moles to grams with clear step-by-step guidance. Explore specific calculations for substances like carbon tetrafluoride and magnesium fluoride. Grasp the nuances of handling different units like millimoles and kilomoles in the context of substances such as carbon disulfide and sulfur trioxide
Want To Learn More? To watch the entire video and ALL of the videos in the series:
full solution
Textbook Solutions (0)
Top Selling Study Tools

Chem 1332, chapter 10, 11 (week 2) (Chemistry)
CHEM 14833
University of Houston
12 pages | Fall 2016